CTLA-4 induced signal transduction in CD8 T-cell differentiation: Implications for tumour-checkpoint therapy
Projektleiter:
Projektbearbeiter:
Aditya Arra,
Dr. rer. nat. Holger Lingel,
Monika Brunner-Weinzierl,
Mandy Pierau
Finanzierung:
Summary of the project:
Tc17 cells are known for low IFN-γ and granzyme B expression, resulting in diminished cytotoxicity. Adoptive transfer into Ag-bearing hosts converts Tc17 cells partially towards IFN-γ producing phenotype still retaining some Tc17 characteristics like increased persistence of survival. Nevertheless molecular mechanisms involved in Tc17 lineage plasticity and stability are yet to be identified. CTLA-4, a homologue to primary co-stimulatory molecule CD28 abrogates T-cell responses by cell intrinsic and extrinsic mechanisms. Studies have shown that CTLA-4 diminishes cytotoxicity of Tc1 cells by selective downregulation of granzyme B, Eomes, and IFN-γ production, but its impact on Tc17 cell differentiation and plasticity is not known.
Introduction:
Optimal T cell activation requires signals via the T cell receptor (TCR) and co-stimulatory molecules. CD28 is the primary co-stimulatory molecule that binds to the B7 ligand molecules and augments and sustains T cells responses. CTLA-4, a homologue to CD28 also recognizes and binds to B7 ligand molecules at a higher affinity than CD28 (1,2). Even though CTLA-4 is known as a negative regulator of T-cell activation, very little is known about its role in regulation of functional responses in T cells.
Results and discussion:
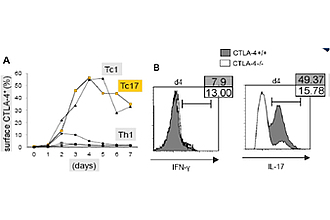
CTLA-4+/+ Tc17 cells produce more IL-17 than CTLA-4-/- Tc17 cells
To evaluate the impact of CTLA-4 on the generation of Tc17 cells, naive CTLA-4+/+ and -/- CD8+ OT.1 T cells were stimulated with OVA257 264 in the presence of Tc17 inducing cytokines. Cytokine expressions were then measured on day3 and 4 when CTLA-4 is expressed on the surface of T cell at a maximum level. CTLA-4+/+ Tc17 cells showed more intracellular IL-17 than CTLA-4-/- Tc17 cells, with a continuous increase from day 2 to the maximum on day 4 after primary stimulation. The distinct difference in IL-17 production was also consistent in co-cultures experiments (equal amounts of CTLA-4+/+ and −/− OT.1 CD8+T cells stimulated with APC), indicating a cell-intrinsic effect of CTLA-4 that enhances differentiation of theTc17 program (3).
Perspectives:
The data demonstrates a cell-intrinsic effect of CTLA-4 that enhances the differentiation of Tc17 cells and role of CTLA-4 in reducing the susceptibility for Tc17 lineage plasticity. Determining the downstream signalling events involved in CTLA-4 mediated Tc17 differentiation and lineage plasticity repression gives an insight into a detailed mechanism of Ipilimumab, a CTLA-4 Mab used in tumour therapy.
Tc17 cells are known for low IFN-γ and granzyme B expression, resulting in diminished cytotoxicity. Adoptive transfer into Ag-bearing hosts converts Tc17 cells partially towards IFN-γ producing phenotype still retaining some Tc17 characteristics like increased persistence of survival. Nevertheless molecular mechanisms involved in Tc17 lineage plasticity and stability are yet to be identified. CTLA-4, a homologue to primary co-stimulatory molecule CD28 abrogates T-cell responses by cell intrinsic and extrinsic mechanisms. Studies have shown that CTLA-4 diminishes cytotoxicity of Tc1 cells by selective downregulation of granzyme B, Eomes, and IFN-γ production, but its impact on Tc17 cell differentiation and plasticity is not known.
Introduction:
Optimal T cell activation requires signals via the T cell receptor (TCR) and co-stimulatory molecules. CD28 is the primary co-stimulatory molecule that binds to the B7 ligand molecules and augments and sustains T cells responses. CTLA-4, a homologue to CD28 also recognizes and binds to B7 ligand molecules at a higher affinity than CD28 (1,2). Even though CTLA-4 is known as a negative regulator of T-cell activation, very little is known about its role in regulation of functional responses in T cells.
Results and discussion:
CTLA-4+/+ Tc17 cells produce more IL-17 than CTLA-4-/- Tc17 cells
To evaluate the impact of CTLA-4 on the generation of Tc17 cells, naive CTLA-4+/+ and -/- CD8+ OT.1 T cells were stimulated with OVA257 264 in the presence of Tc17 inducing cytokines. Cytokine expressions were then measured on day3 and 4 when CTLA-4 is expressed on the surface of T cell at a maximum level. CTLA-4+/+ Tc17 cells showed more intracellular IL-17 than CTLA-4-/- Tc17 cells, with a continuous increase from day 2 to the maximum on day 4 after primary stimulation. The distinct difference in IL-17 production was also consistent in co-cultures experiments (equal amounts of CTLA-4+/+ and −/− OT.1 CD8+T cells stimulated with APC), indicating a cell-intrinsic effect of CTLA-4 that enhances differentiation of theTc17 program (3).
Perspectives:
The data demonstrates a cell-intrinsic effect of CTLA-4 that enhances the differentiation of Tc17 cells and role of CTLA-4 in reducing the susceptibility for Tc17 lineage plasticity. Determining the downstream signalling events involved in CTLA-4 mediated Tc17 differentiation and lineage plasticity repression gives an insight into a detailed mechanism of Ipilimumab, a CTLA-4 Mab used in tumour therapy.
Schlagworte
T-cell, Tumor
Kooperationen im Projekt
Kontakt

Prof. Dr. habil. Monika Christine Brunner-Weinzierl
Otto-von-Guericke-Universität Magdeburg
Experimentelle Pädiatrie und Neonatologie
Leipziger Str. 44
39120
Magdeburg
Tel.:+49 391 6724003
weitere Projekte
Die Daten werden geladen ...


